تاریخ رامینځته شوی ، د پروجیریا د لومړي ځل لپاره د کلینیکي درملو ازموینې کې د هر ماشوم سره د دوی حالت په یو یا ډیرو برخو کې ښه والی ښیې ، دا ثابتوي چې د FTI درملو لونافارنیب د پروجیریا سره د ماشومانو لپاره لومړی پیژندل شوی ، مؤثره درملنه ده.
د کلینیکي آزموینې پایلو کاغذ ستاسو د وړیا کاپي لپاره دلته کلیک وکړئ
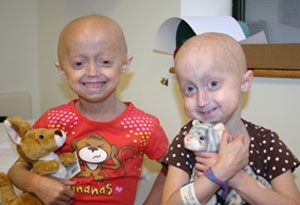
میګن او میغان لومړني دوه ماشومان وو چې په محاکمه کې یې نوم لیکنه وکړه. دوه نجونې په بوسټن کې د دوو کلونو لپاره په هرو څلورو میاشتو کې سره ولیدل، د ازموینې لپاره، د نوي مخدره توکو رسولو لپاره، او یوځای لوبې کول! دوی دلته د 2008 په دسمبر کې د بوسټن د ماشومانو په روغتون کې د ازموینې څخه په وقفه کې دي.
د رسنیو لپاره: دلته کلیک وکړئ د مطبوعاتي اعالمیې، بی رول او نورو مطبوعاتي توضیحاتو لپاره.
د پایلې د ماشومانو لپاره د درملو لومړنۍ کلینیکي آزموینه د پروجیریا سره شتون لري او دا رسمي دی! لونافارنیب، یو ډول فارنیسیل ټرانسفیرس انابیټر (FTI) په اصل کې د سرطان درملنې لپاره رامینځته شوی ، د پروجیریا لپاره مؤثر ثابت شوی. هر ماشوم په څلورو لارو کې په یوه یا ډیرو کې پرمختګ ښیې: اضافي وزن ترلاسه کول، ښه اوریدل، د هډوکي جوړښت ښه کول او/یا، تر ټولو مهم، د وینې رګونو انعطاف زیاتول. د مطالعې پایلې، کوم چې وو د پروجیریا ریسرچ فاؤنڈیشن لخوا تمویل او همغږي شويد سپتمبر په 24، 2012 کې خپور شو د علومو د ملي اکاډمۍ کړنې.
ګورډن ایټ. al.، د هچینسن-ګیلفورډ پروجیریا سنډروم، PNAS، د اکتوبر 9، 2012 vol. 109 شمیره. 41 16666-16671

ناتسوکي، د جاپان څخه، د خپل ورور، پلار او مور سره چې د لونافارنیب - مایع خواږه مخلوط چمتو کوي.
پایلې په ټولو ماشومانو کې د حاصلاتو ښه والی
د شپاړس هیوادونو څخه 28 ماشومانو د 2 ½ کلن مخدره توکو په محاکمه کې برخه اخیستې وه، چې د محاکمې پیل کولو په وخت کې په ټوله نړۍ کې د پروجیریا پیژندل شوي 75 سلنه استازیتوب کوي. ماشومان په هرو څلورو میاشتو کې بوسټن ته سفر کول ترڅو د بوسټن د ماشومانو روغتون له لارې جامع طبي معاینات او درمل مطالعه کړي. د کلینیکي او ژباړې مطالعې واحد. ټولو د اورل لونافرنیب ترلاسه کړ، یو FTI چې د مرک او شرکت لخوا چمتو شوی، د دوو کلونو لپاره په ورځ کې دوه ځله، د کلینیکي آزموینې رییس مارک کیران، MD، Ph.D.، په ډانا کې د ماشومانو د طبي نیورو آنکولوژي رییس تر څارنې لاندې. -فاربر / د ماشومانو د روغتون د سرطان مرکز، او د ډاکټر مونیکا کلینمن او ډاکټر لیسلي ګورډن همکاران
د وزن د زیاتوالي کچه د لومړنیو پایلو اندازه وه، ځکه چې د پروجیریا سره ماشومان په وده کې د سختې ناکامۍ تجربه کوي، د وخت په تیریدو سره د وزن زیاتوالي خورا ورو خطي نرخ سره. څیړونکو د بدن ډیری نورې برخې معاینه کړې، پشمول د شریانونو سختوالی (د زړه د حملې او سټروک وړاندوینه)، د هډوکي سختوالی (د هډوکي پیاوړتیا شاخص) او اوریدل. "کله چې موږ دا کلینیکي محاکمه پیل کړه موږ نه پوهیږو چې ایا د پروجیریا کوم اړخ به د بیرته راګرځیدو وړ وي ، ځکه چې هیڅ چا دمخه د پروجیریا لپاره د کلینیکي درملنې آزموینه نه وه ترسره کړې. موږ وموندله چې د نورو شیانو په مینځ کې ، د وینې لوی رګونه واقعیا وده کولی شي. دا د ماشومانو لپاره یو پرمختللی موندنه وه ، ځکه چې د زړه ګړندۍ ناروغي په پروجیریا کې د مړینې لامل دی. که څه هم د دې لپاره هیڅ لاره نشته چې پوه شو چې ایا موږ د سټروکونو ، د زړه حملې یا د درملنې په دوه کلنه موده کې د اوږدې مودې عمر ډیر کړی دی ، دا مثبتې پایلې موږ دې ته اړ باسي چې نوي درملنې ته دوام ورکړو تر هغه چې موږ هغه څه ترسره کړو چې موږ یې په 1999 کې ترسره کول. موږ غواړو چې د پروجیریا سره ماشومان تر 80 کلونو پورې ژوند وکړي. موږ غواړو چې دوی بشپړ، سالم ژوند وکړي، "ډاکټر ګورډن، د PRF طبي رییس او د درملنې کشف پاڼه لومړی لیکوال وویل.
د پرمختګ سرعت ریکارډ کړئ
د درملنې کشف له یوې لسیزې څخه لږ وروسته د PRF او اوس د روغتیا ملي انسټیټیوټ رییس ډاکټر فرانسس کولینز د پروجیریا لامل پیژندلو لپاره ځواکونو سره یوځای شو - د طبي څیړنې نړۍ کې یو نه اوریدل شوی مهال ویش! مګر د PRF او پروجیریا سره ماشومانو لپاره ، څوک چې یوازې د 13 کلونو اوسط عمر ته ژوند کوي ، دا ډول سرعت د وخت په وړاندې د دې سیالۍ ګټلو لپاره حیاتي دی.
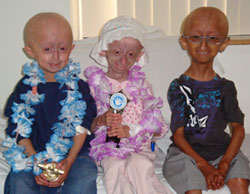
ماتیو، ملاګروس او فرناندو ټول د دوی په وروستي لیدنې کې د PRF څخه د جایزو په ترلاسه کولو موسکا دي. ټرافيان وايي، "تاسو وکړل! تاسو د پروجیریا لومړۍ آزموینه پای ته ورسوله - تاسو یو سوپر سټار یاست!
ډاکټر کیران وویل: "PRF د یوې ادارې یوه ښه بیلګه ده چې په بریالیتوب سره د ژباړې څیړنې فعالوي، د جین کشف څخه کلینیکي درملنې ته په بې ساري سرعت سره حرکت کوي." "له 1999 څخه، کله چې دا سازمان تاسیس شو، تر نن ورځې پورې، PRF د جینیکیک بدلون په ګوته کړی چې د ناروغۍ لامل کیږي، د کلینیکي څیړنې تمویل یې کړی، دا محاکمه یې بشپړه کړې، دویمه آزموینه یې پیل کړې، او اوس مهال د بوستون د ماشومانو په روغتون کې زموږ د ټیم سره کار کوي ترڅو پالن جوړ کړي. د درملو سره یوه بله آزموینه چې د FTIs په څیر ، د پروجیریا حجرو او څارویو ماډلونو کې په زړه پوري پایلې ښودلې. دا د لاسته راوړنې یو په زړه پوری ریکارډ دی.
موږ موافق یو! موږ څنګه دې په زړه پورې ورځ ته ورسیدو؟
د 2003 څخه وروسته د جین بدلون موندنه چې د پروجیریا لامل کیږي، د PRF تمویل شوي څیړونکي پیژندل شوي FTIs د احتمالي درملو درملنې په توګه. د پروجیریا لامل شوی تغیر د پروټین تولید لامل کیږي progerin، کوم چې د حجرو فعالیت ته زیان رسوي. په بدن باندې د پروجیرین زهرجن تاثیر یوه برخه د مالیکول له امله رامینځته کیږي چې د "فارنیسیل ګروپ" په نوم یادیږي ، کوم چې د پروجیرین پروټین سره نښلوي او د بدن حجرو ته زیان رسولو کې مرسته کوي. FTIs د پروجیرین سره د فارنیسیل ګروپ ضمیمه بندولو سره عمل کوي ، د پروجیرین زیان لاملونه کموي.
د مطالعې د نورو جزیاتو لپاره، د مطبوعاتي اعلامیې لپاره دلته کلیک وکړئ
پروجیریا د عمر نورمال پروسې سره تړاو لري
څیړنه ښیې چې د پروجیریا لامل کیږي پروټین progerin په عامو خلکو کې هم پیدا کیږي او د عمر سره زیاتیږي. څیړونکي پلان لري چې د FTIs اغیزې سپړلو ته دوام ورکړي، کوم چې کیدای شي ساینس پوهانو سره د زړه د ناروغۍ په اړه نور معلومات زده کړي چې په میلیونونو اغیزه کوي، او همدارنګه د عمر نورمال بهیر چې موږ ټول اغیزمن کوي.
"د دې نادرې ناروغۍ او نورمال عمر سره نښلول په مهم ډول میوه لري… ارزښتناکه بیولوژیکي بصیرت د نادر اختلالاتو لکه پروجیریا مطالعې سره ترلاسه کیږي. له پیل څخه زموږ احساس دا و چې پروجیریا موږ ته د عمر د عادي پروسې په اړه د زده کړې لپاره ډیر څه درلودل.
- ډاکټر فرانسیس کولینز، د روغتیا ملي انسټیټیوټ رییس

څه موسکا! ماریا په حقیقت کې د بوسټن د ماشومانو په روغتون کې د ازموینې څخه د یوې وقفې په جریان کې د نقاشۍ څخه خوند واخیست.
موږ سره د پروجیریا سره د ټولو ماشومانو موندلو کې مرسته وکړئ نو دوی هم زموږ له کار څخه ګټه پورته کولی شي
څیړونکي پدې باور دي چې په هر وخت کې د پروجیریا سره 200-250 ماشومان ژوند کوي. د نامعلومو ماشومانو پیژندلو لپاره، PRF پیل کړ "نور 150 ومومئ" کمپاین د اکتوبر په 2009 کې، او د 2012 تر سپتمبر پورې، موږ د 96 ماشومانو په اړه پوهیږو چې په 35 هیوادونو کې ژوند کوي - د 83% زیاتوالی!! تاسو کولی شئ د نورو موندلو کې مرسته وکړئ ترڅو دوی د ځانګړي درملنې او پاملرنې څخه ګټه پورته کړي چې PRF چمتو کوي. دا نوي ماشومان ممکن د راتلونکي کلینیکي ازموینې لپاره وړ وي، نو مهرباني وکړئ لاړ شئ نور 150 ومومئ د دې موندلو لپاره چې تاسو څنګه کولی شئ د دې پیښیدو کې مرسته وکړئ.
له ټولو مننه – موږ دا له تاسو پرته نه شو کولی!
یو له اصلي دلیلونو څخه چې موږ پدې لومړۍ آزموینې کې د بریا پایلې ترلاسه کړې د خورا لوی ملاتړ کونکو له امله دي چې تمویل او نور ملاتړ یې چمتو کړی ، زموږ د وروستي هدف ترلاسه کولو ته یو ګام نږدې کیدو کې مرسته کوي - د پروجیریا درملنه. دلته کلیک وکړئ ټولو هغو کسانو ته چې د دې په جوړولو کې یې مرسته کړې ځانګړې خراج تحسین وګورئ خوب د درملنې a حقیقت.
